 International. A research team led by Professor Sangaraju Shanmugam of Energy Science and Engineering at DGIST (Daegu Gyeongbuk Institute of Science and Technology of Korea) has developed a highly efficient and ultra-durable core-shell nanostructured electrocatalyst and successfully replaced the precious anode in water electrolysis, through collaboration with the Pacific Northwest National Laboratory (PNNL) research group, USA.
International. A research team led by Professor Sangaraju Shanmugam of Energy Science and Engineering at DGIST (Daegu Gyeongbuk Institute of Science and Technology of Korea) has developed a highly efficient and ultra-durable core-shell nanostructured electrocatalyst and successfully replaced the precious anode in water electrolysis, through collaboration with the Pacific Northwest National Laboratory (PNNL) research group, USA.
Replacing conventional fuels with renewable energy resources is an appropriate approach to achieving an eco-friendly environment and decreasing future energy demands. For these purposes, the generation or conversion of electrochemical energy into renewable energy devices, which depends on anode and cathode reactions, has received a lot of attention.
In electrocatalytic water splitting, oxygen gas is generated at the anode due to the oxygen evolution reaction (OER), and is a slow electrochemical reaction compared to the hydrogen evolution reaction (HER). Therefore, a suitable electrocatalyst is needed for promising and stable electrocatalytic water splitting.
The development of efficient, durable and low-cost OER electrocatalysts is a big challenge and paid more attention to the renewable energy devices of the water electrolyzer. Until now, ruthenium and iridium oxides are considered state-of-the-art electrocatalysts in OER, but the lack of stability limits their use in large-scale water division and makes widespread commercialization difficult.
Therefore, The teams of Professor Shanmugam and PNNL have focused on developing an alternative, low-cost non-noble metal electrocatalyst to replace the noble metal anode electrode in an efficient water division. Carbon-supported metal has been considered as an efficient electrocatalytic material for improved OER in water splitting. So far, most of the electrocatalysts developed have had a higher carbon content and less specious content of active metals. The increased amount of carbon flooded the active sites of real metal and thus resulted in faster carbon corrosion conditions. This led to lower electrocatalytic activity, stability, and large-scale water splitting processes (scalability).
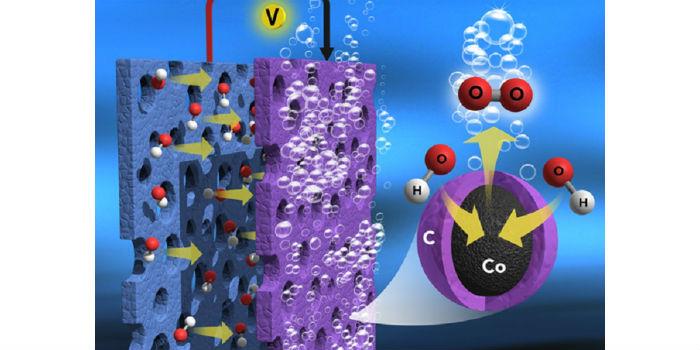
In the study, the researchers found that a large number of inorganic cobalt metal ions bonded by organic ligands in the Prussian blue analogue proved to be a suitable precursor for the development of nitrogen-doped, efficient and ultrastable graphitic nanocarbon, rich in encapsulated core and layer electrocatalyst metals for the OER (anode) inactive in water splitting.
When heated (600-900 °C) in an inert atmosphere, the cobalt metal ions and organic ligands in the salt are transformed into thin carbon layers of nitrogen-doped graphite and cobalt metal, respectively, which form cobalt,thin-layered cobalt and encapsulated metal core-shell nanostructures (Core-Shell Co NC). The thin layers of carbon have a strong interaction with the cobalt metal, which can promote less carbon corrosion, excellent electron movement, and more exposure of cobalt metal to the reaction medium, including the formation of nanometric morphology without particle aggregation.
Consequently, the combined effect of carbon and cobalt metal on the electrodes achieves a more efficient electrocatalytic activity of the OER than that of precious metal electrodes to allow for efficient water splitting. Therefore, the metal-rich non-noble electrode is an alternative, active, stable and less expensive OER anode for the cost-effective production of H2 gas in commercial-scale water electrolysis.
Source: Daegu Gyeongbuk Institute of Science and Technology of Korea.

























Leave your comment